
All anchors associated with Abbott SCS MR. owns the largest amount of shares in Abbott Laboratories (155.63M shares representing 8.95% of the company), but there are many stakeholders in the company. Model 3660 / 3662 (with Penta 5-column paddle lead, 60cm, Model 3228) The implantable pulse generator (IPG) for the Proclaim XR SCS System allows for MR Conditional scans of any body part, head, or extremity when the IPG is coupled with the correct lead, and implanted in an approved location. 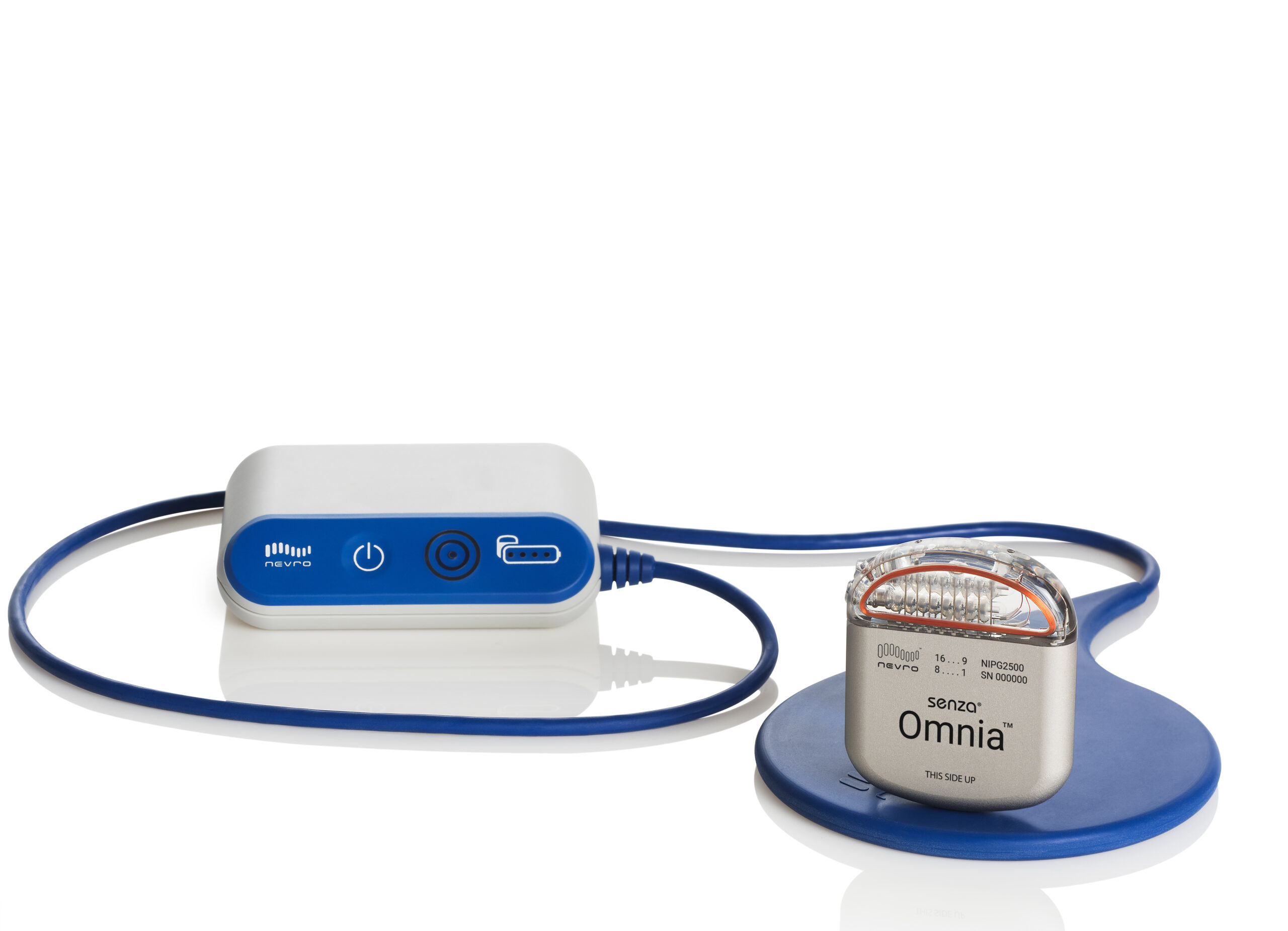
This neurostimulation system is contraindicated for patients who are unable to use the system. Refer to the Indications for Use Data Sheet for Abbott Medical spinal cord stimulation systems. Who Owns Abbott Medical Devices?Ībbott was originally founded by Chicago physician Wallace Calvin Abbott in 1888. Abbott Medical-approved trial leads and ex tensions to deliver trial stimulation for a maximum of 30 days. The Pain Center utilizes Abbott’s neuromodulation devices that treat chronic pain, movement disorders, and other neurological conditions, including spinal cord stimulation systems and deep brain stimulation systems. Glucose monitoring systems (e.g., Abbott Freestyle Libre).They make a wide range of medical devices with some of the most well-known products, including the following: Hassle-free means recharge-free.Abbott creates professional products that focus on cardiovascular and diabetes care, diagnostics, neuromodulation, nutrition, and medicines. Refer to the IFU for additional information. Safety comparisons and specific dose-response curves for each dosage have not been clinically established. An electrode: this sits in the epidural space and delivers very small and precise currents. It is implanted under the skin and has an inbuilt battery. NOTE: In neurostimulation therapy, ‘dose’ refers to the delivery of a quantity of energy to tissue. A spinal cord stimulator system is made of four parts: Impulse Generator (IPG): this is a computer that is roughly the size of a matchbox that controls the impulses delivered to the spinal cord. *Up to 10 years of battery longevity at the lowest dose setting: 0.6mA, 500 Ohms, duty cycle 30s on/360s off. 
The Abbott Proclaim XR SCS System benefits include Model 3660 / 3662 (with Octrode stimulation lead, 60cm, model 3186) The implantable pulse generator (IPG) for the Proclaim XR SCS System allows for MR Conditional scans of any body part, head, or extremity when the IPG is coupled with the correct lead, and implanted in an approved location.No use of any Abbott trademark, trade name, or trade dress in this site may be made without the prior written authorization of Abbott, except to identify the product or services of the. The Abbott Proclaim XR can seamlessly fit into a patient’s lifestyle by allowing them to discreetly manage their pain by using familiar Apple mobile digital devices and Bluetooth wireless technology. Unless otherwise specified, all product names appearing in this Internet site are trademarks owned by or licensed to Abbott, its subsidiaries or affiliates. Abbott’s Proclaim XR spinal cord stimulation device was first approved to treat chronic pain in 2019. 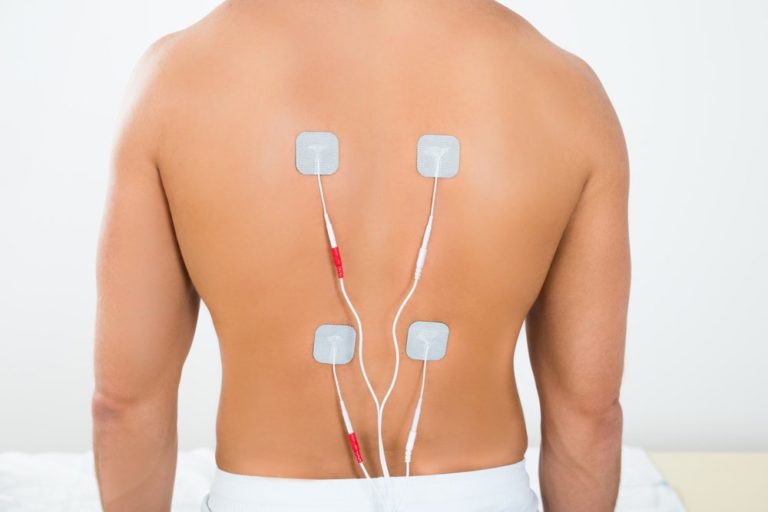
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
